Mitovation is developing proprietary infrared light (IRL) technology for the treatment of ischemia-reperfusion injuries of the brain. Ischemia-reperfusion brain injuries are an important medical problem because they can lead to death or irreversible medical problems that greatly affect a person’s neurological function and quality of life. In adults, cardiac arrest, traumatic brain injury, or stroke can cause cognitive or functional impairments that are either irreparable or take months to years of therapy to recover. In children and newborns, cardiac arrest, traumatic brain injury, and emergent medical events like Hypoxic Ischemic Encephalopathy (HIE) can not only lead to functional and cognitive impairments, but also to devastating medical conditions like cerebral palsy. Today, there are no FDA-approved treatments for neurological ischemia-reperfusion injury.
Understanding Reperfusion Injury

Healthy MitochondriaMitochondria consume oxygen to produce energy
Mitochondria are the “power plants” of cells. In a cell, healthy mitochondria generate energy to drive many subcellular processes.

IschemiaIschemia creates cellular stress due to lack of oxygen
Reperfusion injury is additional cellular/tissue damage caused by restoration of normal blood flow to ischemic tissue. This restoration of circulation results in oxidative stress (free radical generation), programmed cell death, inflammation, and further damage to the affected tissue.

Reperfusion without Mitovation TherapyReperfusion injury leads to further cell death
Lack of oxygen and nutrients during a period of zero blood flow creates an unstable and vulnerable cellular environment. Restoration of normal circulation to these cells results in oxidative stress, apoptosis (programmed cell death), inflammation, and further tissue damage to the affected area.

Reperfusion with Mitovation TherapyMitovation’s therapy limits oxidative stress
Mitovation’s unique technology addresses ischemia-reperfusion injury by minimizing oxidative stress to brain tissue during this critical period in patient care. The combination of patent-protected inhibitory IRL wavelengths used in Mitovation’s non-invasive medical device, together with Mitovation’s waveguide technology (patent pending) that enables the safe delivery of a therapeutic dose of IRL deep into the brain, modulate subcellular reactions in mitochondria to prevent cell death and produce a neuroprotective effect.

Small Animal Study Outcomes
Research that began in the labs of Drs. Maik Hüttemann and Thomas Sanderson led to the discovery of certain wavelengths of IRL that can have a neuroprotective effect on cells undergoing reperfusion after an ischemic event. These novel findings that began on a research bench gave way to further exploration of the technology’s potential with the creation of a prototype medical device and the advent of small animal trials (rodent). Rodent study outcomes were overwhelmingly positive with IRL treatment groups displaying better overall survival, less brain tissue damage, and higher cognitive function than untreated control groups.
In control groups from small animal studies, cardiac arrests/resuscitation (CA/CPR) with no Mitovation therapy resulted in severe disability or death. Histologic evaluation of hippocampus tissue in survival studies showed extensive loss of neurons 4 days after initial CA/CPR in Controls. In contrast, Mitovation-treated animals exhibited near-normal neurologic function post CA/CPR. Histologic samples of hippocampus tissue from Mitovation-treated animals showed greater preservation of neurons in this region and exhibited reduced neurological deficits compared to the Control group.

Sham
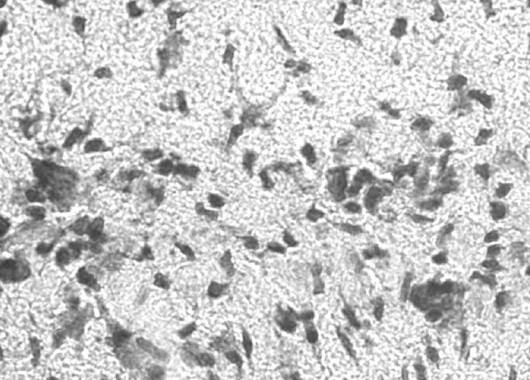
Control
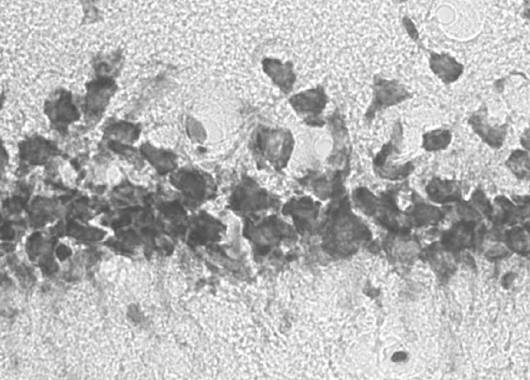
Mitovation

Large Adult Animal Study Outcomes
After establishing proof of concept in small animal models of cardiac arrest and resuscitation, Drs. Hüttemann and Sanderson received grant funding from the NIH and DoD to investigate their technology’s potential in large animal models (pig). Data collected from large animal models of adult cardiac arrest confirmed that IRL has a neuroprotective effect on cells undergoing ischemia-reperfusion injury. The Neurologic Deficit Score (NDS) graph shows the NDS of Sham (no cardiac arrest, no Mitovation treatment), Control (cardiac arrest/resuscitation with no Mitovation treatment), and Mitovation treated groups (cardiac arrest/resuscitation with Mitovation therapy). Mitovation treated groups showed significantly better neurological outcomes compared to the untreated Control group.
In control groups from adult large animal studies, cardiac arrests/resuscitation (CA/CPR) with no Mitovation therapy resulted in severe disability or death. Histologic evaluation of hippocampus tissue in survival studies showed extensive loss of neurons 4 days after initial CA/CPR in Controls. In contrast, Mitovation-treated animals exhibited near-normal neurologic function post CA/CPR. Histologic samples of hippocampus tissue from Mitovation-treated animals showed greater preservation of neurons in this region and exhibited reduced neurological deficits compared to the Control group.

Sham

Control

Mitovation
Large animal neonatal study
Drs. Hüttemann and Sanderson investigated the use of Mitovation’s technology in a large animal model (pig) of neonatal HIE - this work was supported by an ongoing NIH small business (STTR) grant. Data from this study also showed Mitovation-treated neonates had significantly better neurological outcomes, fewer seizures, and less damage to brain white matter compared to the untreated Control group.

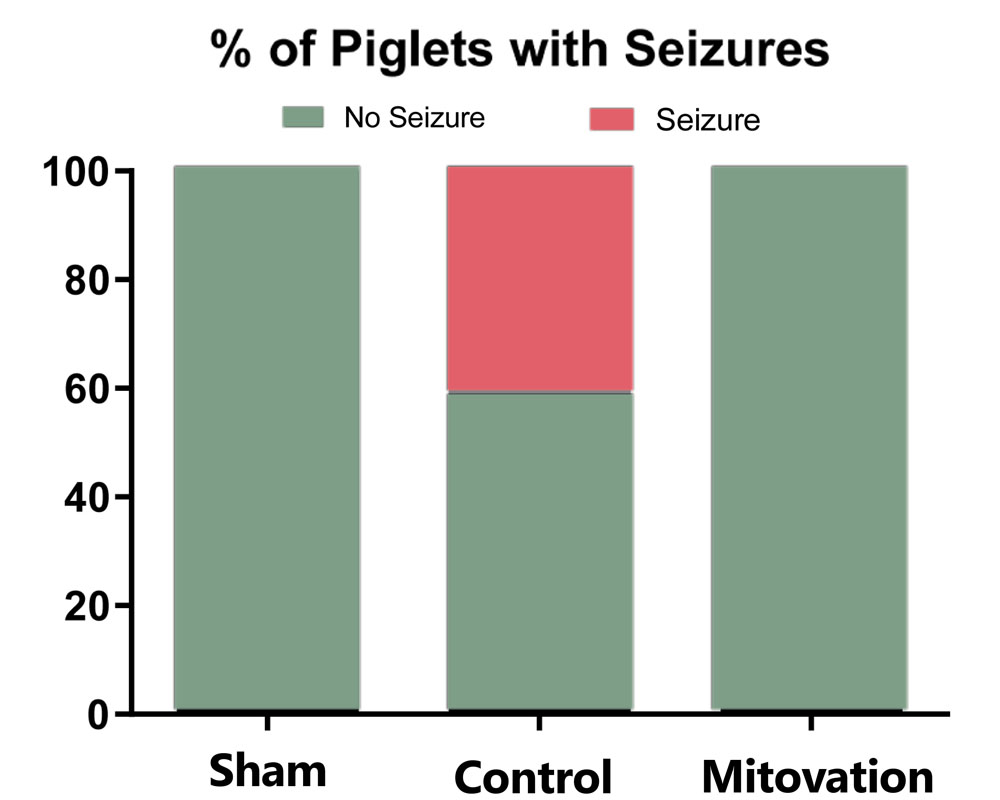

In control groups from neonatal animal studies (pig), cardiac arrests/resuscitation (CA/CPR) with no Mitovation therapy resulted in severe disability, seizures, or death. Histologic evaluation of cortical white matter in survival studies showed extensive loss of brain white matter 14 days after initial CA/CPR in Controls. In contrast, Mitovation-treated animals exhibited near-normal neurologic function post CA/CPR. Histologic samples of cortical white matter from Mitovation-treated animals showed greater preservation of neurons in these regions and exhibited reduced neurological deficits and seizures, with better overall survival.
Normal Brain

Sham
Severe White Matter
Damage
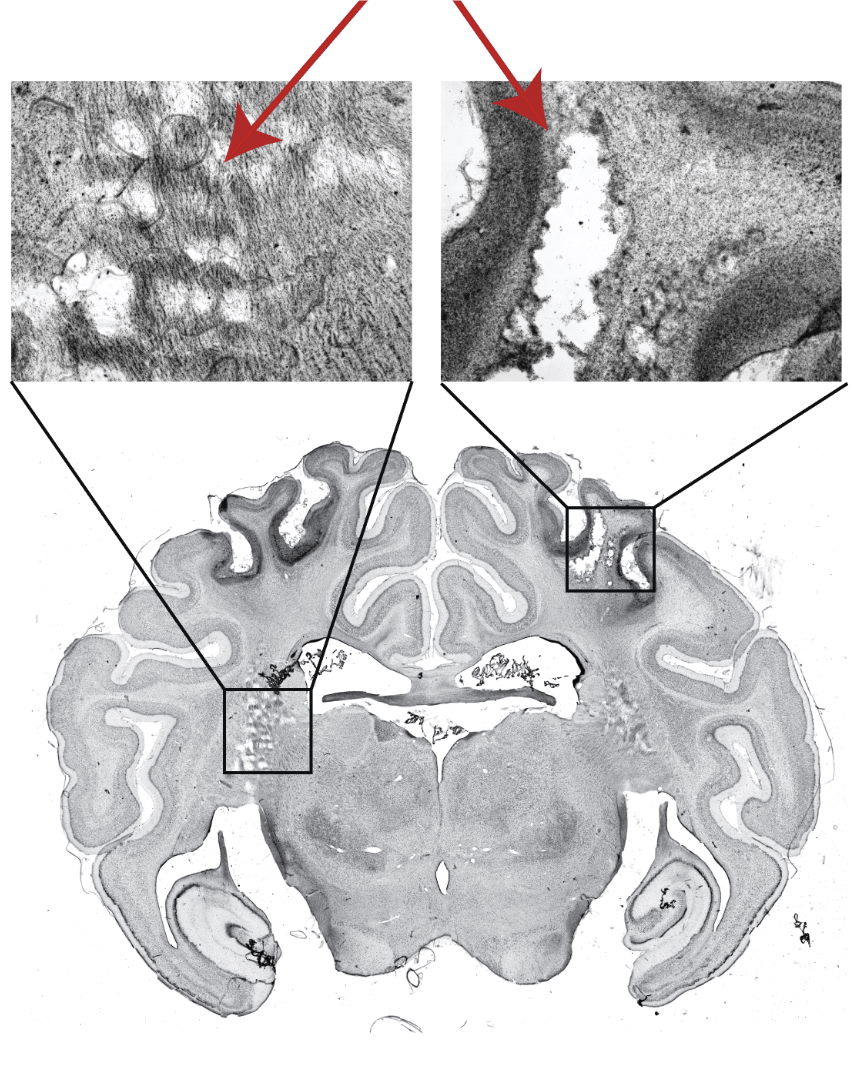
Control
Significantly Reduced
Damage

Mitovation
Patent Protected Technology
with First in Class Design
Mitovation has partnered with ISO 13485 certified and FDA compliant medical device firms for the design, engineering, and manufacture of its devices for human use.
What's Next?
Mitovation is currently focused on confirming our previous findings with ongoing grant support from the NIH and others, designing a human interface for its medical device, and preparing for first-in-human clinical trials in pursuit of FDA approval. We believe our technology is poised to change the standard of care for those who suffer from ischemia-reperfusion injuries of the brain.Located in Ann Arbor, MI. Get in touch to schedule a meeting
Let's TalkRecent Publications
To read more about Mitovation’s technology and its mechanism of action, please click here to read our recent publications.Citations
(1) Benjamin EJ, Muntner P, Alonso A, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. 2019;139(10):e56-e528.
(2) Kurinczuk JJ, White-Koning M, Badawi N. Epidemiology of neonatal encephalopathy and hypoxic-ischaemic encephalopathy. Early Hum Dev. 2010;86(6):329-38.

